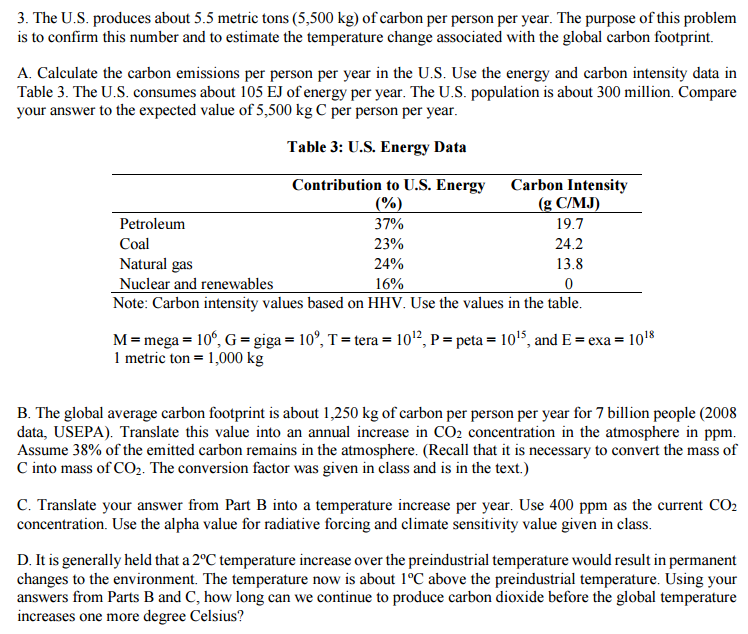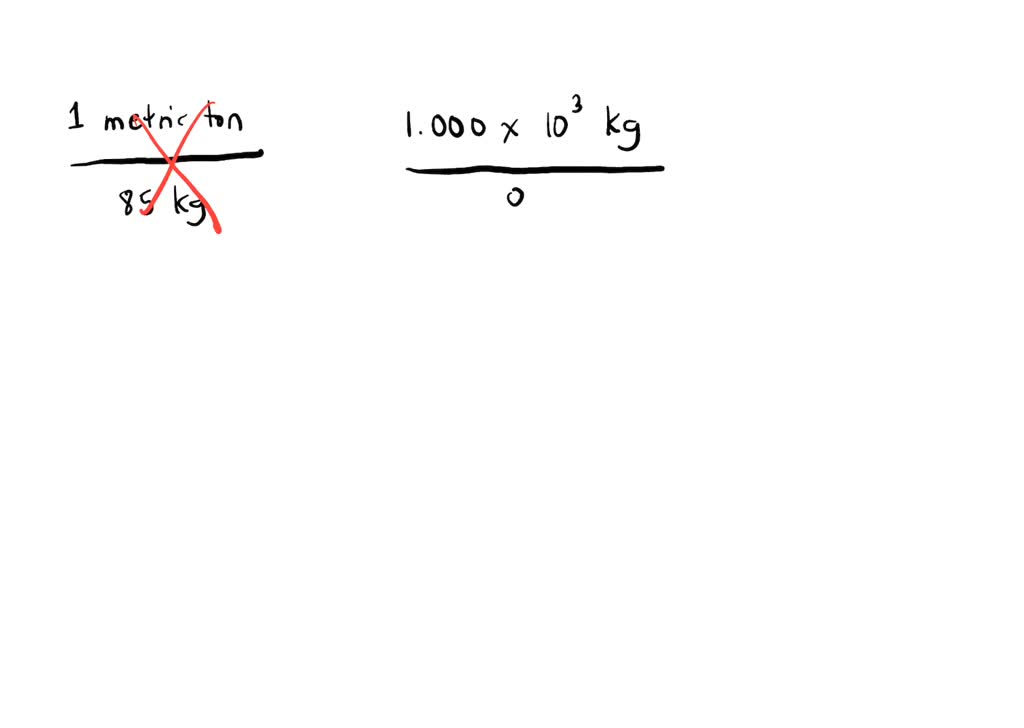
SOLVED:If a metric ton is 1.000 \times 10^{3} \mathrm{kg}, how many 85 \mathrm{kg} people can safely occupy an elevator that can hold a maximum mass of exactly 1 metric ton?

SOLVED:In the U.S. Customary System of measurement, 1 ton =2000 Ib. In the metric system, 1 metric ton =1000 \mathrm{kg}. Use this information to answer Exercises 69-72. A Lincoln Navigator weighs 5700

Calculate the number of moles in the following masses: (i) 1.46 metric tones of Al (1 metric ton =10^(3) kg ) (ii) 7.9 mg of Ca