Full article: Pharmacokinetic bioequivalence, safety and acceptability of Ornibel®, a new polymer composition contraceptive vaginal ring (etonogestrel/ethinylestradiol 11.00/3.474 mg) compared with Nuvaring® (etonogestrel/ethinylestradiol 11.7/2.7 mg)

Color, Scent and Size: Exploring Women's Preferences Around Design Characteristics of Drug-Releasing Vaginal Rings | SpringerLink

Progesterone vaginal ring as a new contraceptive option for lactating mothers: Evidence from a multicenter non-randomized comparative clinical trial in India - Contraception

PDF) Pharmacokinetic bioequivalence, safety and acceptability of Ornibel ® , a new polymer composition contraceptive vaginal ring (etonogestrel/ethinylestradiol 11.00/3.474 mg) compared with Nuvaring ® (etonogestrel/ethinylestradiol 11.7/2.7 mg)
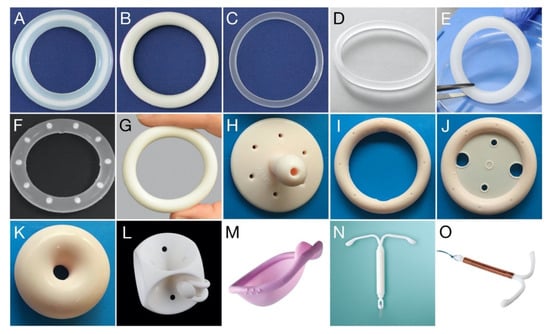
Pharmaceutics | Free Full-Text | The Vaginal Microbiota, Bacterial Biofilms and Polymeric Drug-Releasing Vaginal Rings | HTML